FPS Receives Approval for HTS- and PCR-Based Diagnostic Protocols for Quarantine Release and Certification of Grapevine, Prunus and Roses
The US Department of Agriculture-Animal and Plant Health Inspection Service, Plant Protection and Quarantine (USDA-APHIS-PPQ) and California Department of Food and Agriculture (CDFA) have approved the use of Foundation Plant Services’ (FPS) revised diagnostic testing protocol that replaces biological indexing with a combination of high throughput sequencing (HTS) and polymerase chain reaction (PCR) testing for release of plant material. Research conducted at FPS indicates that these methods used in combination result in more accurate test results compared to biological indexing using woody and herbaceous indicators for virus detection. In turn, this will safeguard imported plant material ensuring detection of quarantine pathogens of concern and potential unknown pathogens. Furthermore, adoption of the streamlined testing methods will yield the most accurate information about the phytosanitary status of incoming material, expedite release times, and reduce potential risks from the transmission of vector-mediated viruses in the field.
Registration and Certification (R&C) Programs, such as those for grapevines and fruit and nut trees administered by CDFA, target the elimination of specific viruses that are spread through grafting or propagation. Plants destined to enter the CDFA R&C Programs are tested, or are propagated from sources that have been tested, using methods prescribed in the CDFA regulations for foundation stock. These methods include herbaceous and woody indexing in addition to molecular testing methods such as PCR. Biological indexing using woody and herbaceous indicators has been the gold standard method for certification programs for decades.
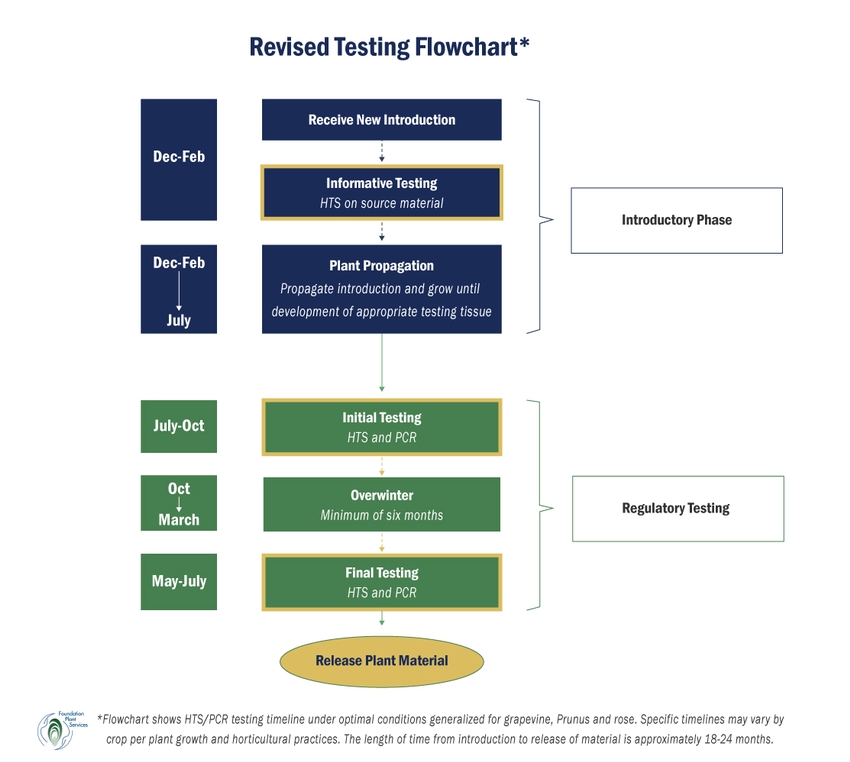
Since 2007, FPS has been a leader in adopting the use of HTS technology for plant diagnostics. HTS is an alternative to traditional diagnostic methods for the detection and identification of organisms in a sample by using DNA sequencing to provide a comprehensive picture of the entire microbial profile. FPS scientists have conducted side-by-side studies comparing the efficacy of woody and herbaceous indexing to PCR and HTS testing in grapevine, Prunus, and rose diagnostics. The results of these studies indicate that biological indicators give false negative results a significant percentage of the time and do not provide sufficient sensitivity in detecting target viruses or unknown viral pathogens. Similar results were obtained in studies conducted on Prunus and pome fruits by other clean plant centers. In addition, biological indexing requires a minimum of two to four years before plant material can be released. FPS has integrated this research knowledge into an improved protocol that adopts the most reliable and efficient techniques using HTS and real time quantitative RT-qPCR/qPCR, which are known to be highly sensitive tests. The revised protocol (see testing flowchart below) uses informative HTS testing of source material to provide customers with feedback on the phytosanitary status of the plant material and determines if virus elimination therapy is necessary. Once plants are propagated and have developed appropriate tissue to test, initial HTS/PCR testing provides information on viruses that may be present in the plant material in the summer to fall. Final HTS/PCR testing after a six-month minimum period of dormancy provides a third and final test, testing for the presence of viruses in the spring to summer and verifying earlier test results or detecting pathogens previously undetected due to low titer. The entire process captures pathogens that may be present in plant source material by testing upon arrival, then in two different seasons separated by a period of dormancy to account for fluctuations in virus titer. Although there are slight variations between the grapevine, Prunus, and rose protocols per the horticultural needs of each crop, the length of time from introduction to release of material using the revised protocol is approximately 18-24 months.
Research demonstrating the accuracy and benefits of the use of a combination of PCR and HTS testing in lieu of biological indexing to evaluate viruses and virus-like organisms has led to USDA-APHIS and CDFA adoption of a revised testing protocol for plant material entering the FPS program. In 2020, FPS was granted approval by CDFA to replace all biological indexing with a combination of PCR and HTS testing for the Fruit & Nut Tree and Grapevine R&C Programs. In August 2021, USDA-APHIS discontinued the biological indexing requirement and approved a combination of PCR and HTS testing for Vitis spp., Prunus spp. and Rosa spp. accessions that are sourced under a PPQ 588 Controlled Import Permit. This approval also applies to other clean plant centers using a protocol with a combination of HTS and PCR testing in lieu of biological indexing.
There are additional benefits to eliminating reliance on field indicators. First, maintaining quarantine material in an unsecure field location during the lengthy field indexing period presents a risk due to the potential transmission of unknown, exotic vector-meditated viruses. Discontinuing biological indexing in favor the more secure, reliable, and efficient methods of PCR and HTS testing reduces this risk. Second, HTS is an excellent method for detecting diseases of unknown etiology as well as known viruses. Since incorporating HTS in plant virus diagnostics, FPS has accumulated additional data on virus diversity. Incorporation of this data into assay design increases PCR detection ability. Third, FPS will continue working closely with other clean plant centers in the US to harmonize testing protocols and these protocol changes are in-line with the International Plant Protection Convention recommendations. This allows the US to collaborate internationally and operate within the European framework. Finally, the current time required for plant introductions to move through the pipeline is lengthy. The entire process from introduction to release of material using biological indicators ranges from two to six years. The revised protocol can reduce the regulatory testing process to one-year, excluding the time required to establish plants. This expedited timeline with quick access to clean plant material is critical for certification programs and enables the US industry to remain competitive and responsive to consumer needs.
Foundation Plant Services wishes to thank: Clean Plant Center Northwest; USDA APHIS PPQ Plant Germplasm Quarantine Program; USDA APHIS PPQ-PHP-IRM, Plants for Planting Policy; CDFA Plant Health and Pest Prevention Services; CDFA Nursery Services program; the National Clean Plant Network and industry leaderships who contributed to the important improvements to the standard operating procedures and the approval of the HTS- and PCR-based diagnostic protocol.