Foundation Plant Services (FPS) has long been a source of elite propagation materials of commercially important Prunus scion and rootstock cultivars as part of the California Department of Food and Agriculture’s (CDFA) Deciduous Fruit and Nut Tree Registration and Certification Program. In 2016, FPS acquired a Controlled Import Permit (P588) from the United States Department of Agriculture, Animal Plant Health Inspection Service, Plant Protection and Quarantine (USDA APHIS PPQ) to facilitate the introduction, quarantine, and release of imported Prunus for the fruit tree industry. Recent advances in technology and updates to testing requirements have led to the release of 20 Prunus selections on an accelerated timeline.
In addition to molecular testing, USDA APHIS PPQ prescribed procedures have required the use of biological indicators to assess the disease status of quarantine material. In the past, regulations have required the grafting of candidate selections to four Prunus biological indicators (P. persica ‘GF 305’, P. avium ‘Canindex’, P. avium ‘Bing’, and P. serrulata ‘Kwanzan’) that were observed for symptom development in a greenhouse for at least one full growing season. If no disease symptoms were observed, the selection might be eligible for full quarantine release. This process frequently needed to be repeated if the imported selection underwent virus therapy, sometimes adding years to the quarantine process.
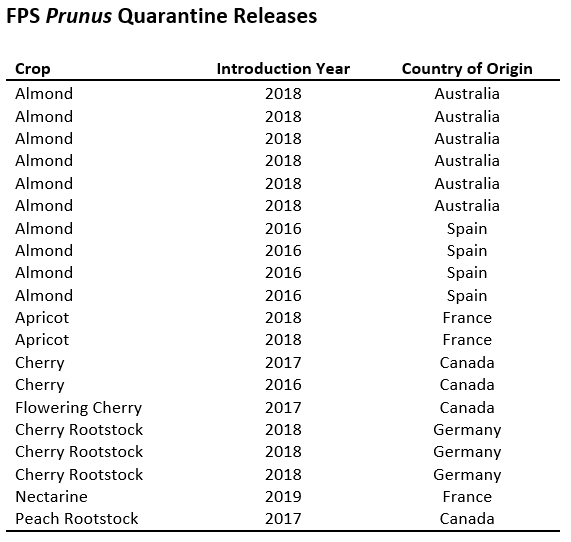
Dr. Maher Al Rwahnih, Diagnostic and Research Laboratory Director at FPS and a leader in high throughput sequencing (HTS) use in plant diagnostics, recognized the potential of applying the advantages of HTS technology to the fruit tree quarantine testing requirements. HTS has advantages over traditional diagnostic methods as it is sensitive, rapid, comprehensive, and able to discover even previously unknown viruses. Dr. Al Rwahnih’s analysis of the results of biological indexing in a side-by-side comparison to modern laboratory diagnostic methods revealed that the performance of the biological indicators was inferior. Furthermore, since reliable laboratory tests were available to test for known pathogens detected by Canindex and Kwanzan, the removal of these two indicators that were redundant to the PCR and HTS tests was proposed. A new protocol for fruit tree introduction and testing was developed in coordination with Dr. Scott Harper at the Clean Plant Center Northwest (Prosser, Washington) to adopt the new testing approach and harmonize the standard operating procedures of the two Prunus quarantine programs. USDA APHIS PPQ and the CDFA approved the new protocol with the removal of Canindex and Kwanzan as biological indicators. Under the new protocol, plants are subjected to greenhouse indexing and molecular testing at two different time points by PCR and HTS. Plants may be released if all test results demonstrate plants are free of viruses or virus like agents. The culmination of this work and collaboration is the release of 20 selections (see table), with an anticipated release of 11 additional selections in late summer 2020. The full release of new plant material under the new protocol will only take eight months to one year if it enters the program free of target viruses. Fruit tree nurseries and growers will greatly benefit from the release of material under this protocol as highly anticipated material be available to nurseries sooner, allowing them to begin their propagation and distribution of quality material to growers.
Foundation Plant Services wishes to acknowledge the work of Clean Plant Center Northwest; USDA APHIS PPQ-PHP-IRM, Plants for Planting Policy; USDA APHIS PPQ Plant Germplasm Quarantine Program; CDFA Plant Health and Pest Prevention Services; CDFA Nursery Services program, and the National Clean Plant Network leadership who all contributed to this important improvement of the standard operating procedures for fruit tree quarantine material leading to the release of these selections.